Hydrolysis of ATP
May 17, 2024 | by Bloom Code Studio

It is the catabolic reaction process where the energy-rich phosphodiester bonds of ATP molecules are broken down (hydrolyzed), releasing energy and inorganic phosphate molecules in the presence of water and ATPase enzyme. It is an exergonic reaction where the energy stored in the phosphodiester bond during ATP formation is released. This released energy is used by the cell for performing several cellular activities and reactions.
ATP is first hydrolyzed, breaking one energy-rich phosphodiester bond to form ADP. The ADP molecule can further be hydrolyzed breaking another energy-rich phosphodiester bond to form AMP. The breakdown of phosphodiester bond is catalyzed by ATP hydrolase (ATPase) enzyme in presence of water. ATP hydrolysis is a reversible reaction i.e., ADP and AMP can be rephosphorylated from ATP molecule.
Hydrolysis of ATP to ADP releases 7.3 kCal/mol of energy. It can be expressed as:
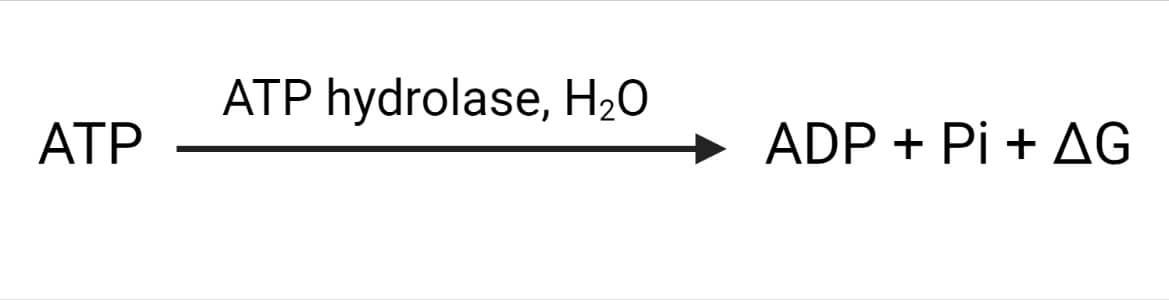
Where, ∆G= Gibbs free energy = – 7.3 kCal/mol energy
Further hydrolysis of ADP to AMP releases 7.5 kCal/mol of energy. It can be expressed as:
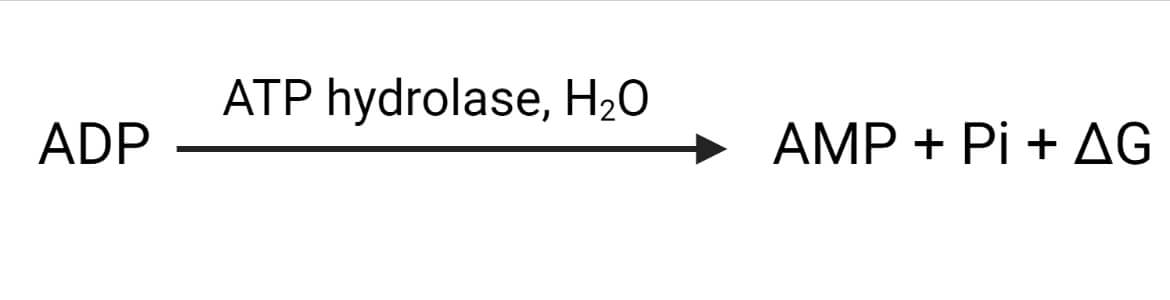
Where, ∆G= Gibbs free energy = – 7.5 kCal/mol energy
The overall reaction can be summarized as:
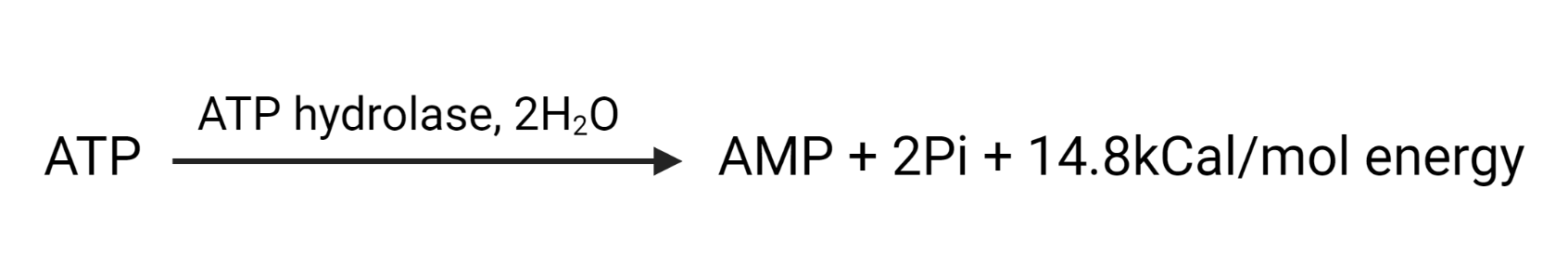
RELATED POSTS
View all
