Osmotic Pressure
May 13, 2024 | by Bloom Code Studio

- Osmotic pressure is the pressure applied by a hypotonic pressure that results in the movement of solvent molecules across the semi-permeable membrane.
- It is the minimum pressure that should be applied to the solution to prevent the inwards flow of pure solvent across the semi-permeable membrane.
- Osmotic pressure is the driving force of osmosis and the rate of osmosis increases as the osmotic pressure increases.
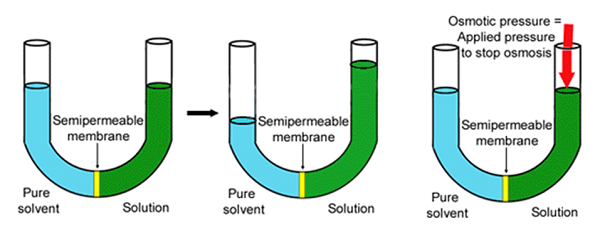
The osmotic pressure of a solution can be calculated as follow:
∏= MRT
Where ∏ is the osmotic pressure
M is the molar concentration of the solute
R is the gas constant
And T is the temperature of the system.
RELATED POSTS
View all
