Do you know what the allotrope of carbon means?
April 28, 2024 | by Bloom Code Studio

Allotrope is nothing but the existence of an element in many forms which will have different physical properties but will have similar chemical properties and its forms are called allotropes of allotropic forms. Allotropes are defined as the two or more physical forms of one element. These allotropes are all based on carbon atoms but exhibit different physical properties, especially with regard to hardness.
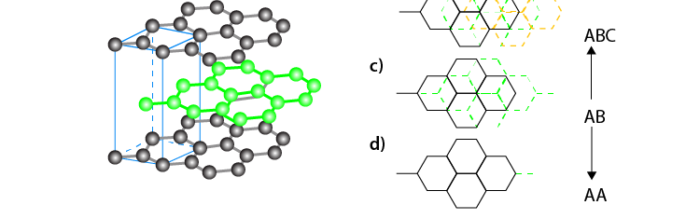
The common, crystalline allotropes of carbon are Diamond, Graphite, Graphene, Fullerene ( C60), and Carbon nanotubes. Carbon shows allotropy because it exists in different forms of carbon such as diamond, and graphite. But it is now known that all the amorphous carbons contain microcrystals of graphite. Though these allotropes of carbon have different crystal structures and different physical properties, their chemical properties are the same and show similar chemical properties. Both diamond and graphite have the symbol C. Both give off carbon dioxide when strongly heated in the presence of oxygen.
The structure of carbon allotropes are
Diamond
Diamond is a well known allotrope of carbon having a three-dimensional network structure. It exhibits the highest hardness and thermal conductivity.

Graphite
Graphite is another allotrope of carbon. It is a two dimensional structure and a good conductor of electricity.
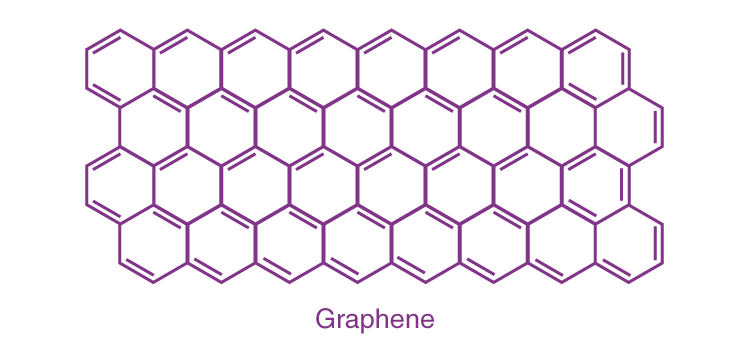
Graphene
It is a single layer allotrope of carbon having a two dimensional honeycomb structure. Graphene has very high electron mobility and, like graphite, is a good electrical conductor, due to the occurrence of a free pi (p) electron for each carbon atom.
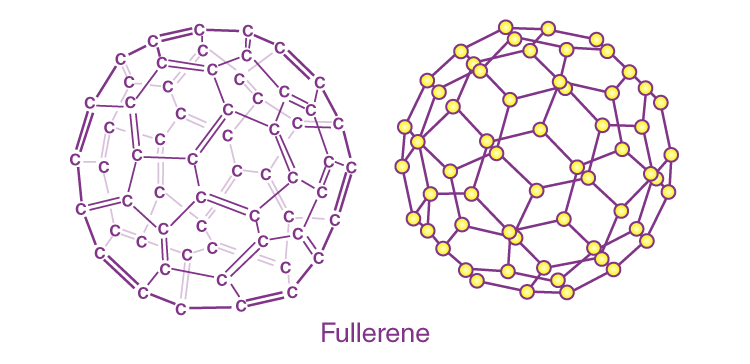
Fullerene (C6O)
A fullerene is an allotrope of carbon atoms connected by single and double bonds to form a closed or partially closed structure, with fused rings of five to seven atoms.
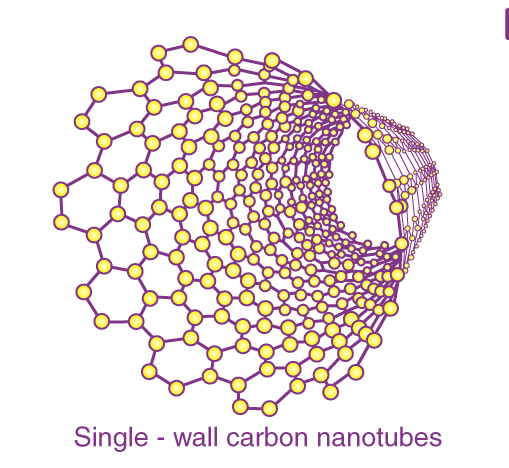
Single-wall carbon nanotubes
Single-wall carbon nanotubes are one of the allotropes of carbon, intermediate between fullerene cages and flat graphene.
RELATED POSTS
View all
