
What are Monatomic Gases?
“Monatomic” is a combination of two words “mono”, and “atomic” means a single atom. This term is used in both Physics and Chemistry and is applied to the gases as monatomic gases. In the gaseous phase at sufficiently high temperatures, all the chemical elements are monatomic gases.
Noble gases are monatomic gases as they are unreactive, which is a property of these gases. They do find applications in daily life like
- Helium is used in filling balloons as their density is lower than the air’s.
- Neon is used for creating advertising signs as they glow when electricity flows through them.
- Argon is used in a light bulb to prevent the burning of the filament as it is unreactive.
What are Diatomic Molecules?
Diatomic molecules are those molecules that are composed of only two atoms. If a diatomic molecule is composed of the same element, it is known as a homonuclear, and if it is composed of two different elements, it is known as heteronuclear.
What is a Polyatomic Ion?
A polyatomic ion is also known as a molecular ion that is composed of two or more covalently bonded atoms. It is also referred to as a radical.
Monatomic Examples
The noble gases are examples of monatomic gases, and they are:
- Helium
- Radon
- Neon
- Xenon
- Argon
- Krypton
Diatomic Molecules Example
Diatomic elements are non-metals and exist in pure form as:
| Hydrogen H2 |
| Nitrogen N2 |
| Fluorine F2 |
| Oxygen O2 |
| Iodine I2 |
| Chlorine Cl2 |
| Bromine Br2 |
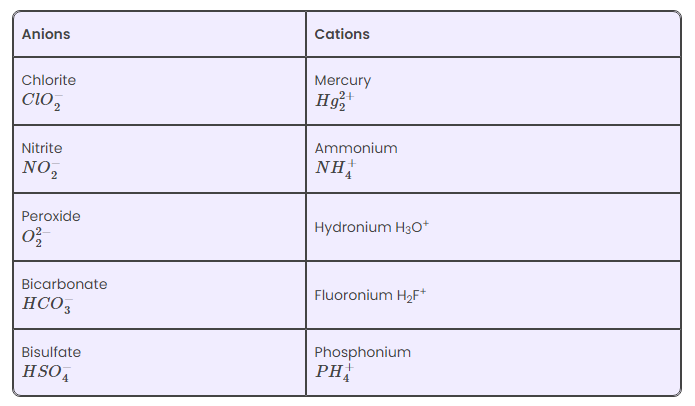
Examples of Polyatomic Ions
Following is the common list of polyatomic ions:
RELATED POSTS
View all
