
We saw the intermediates that could be formed in an organic reaction; now, let us look into transition states and the difference between an intermediate and a transition state.
The intermediates in organic chemistry are formed in a multi-step reaction, but some reactions can occur in a single step without having to form an intermediate. These reactions will occur by going through a transition state. This can be clear by looking at the energy profile diagram for a reaction, R→P.
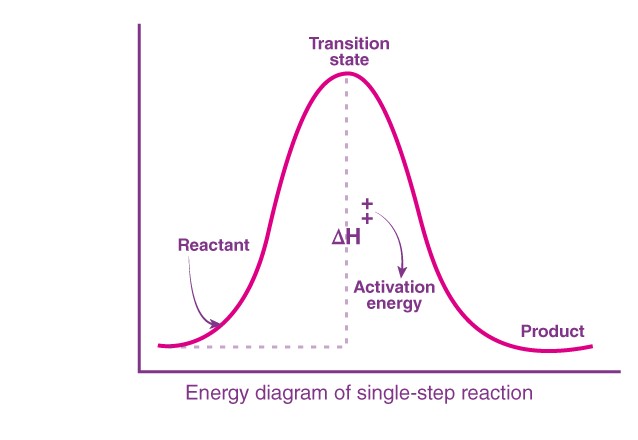
The transition state corresponds to the highest energy in the reaction, after which it can give either the products or, in the case of a reversible reaction, the reactants.
Consider a reaction, A→D with the following steps, A→B, B→C, and C→D
The energy profile for this reaction is given below:
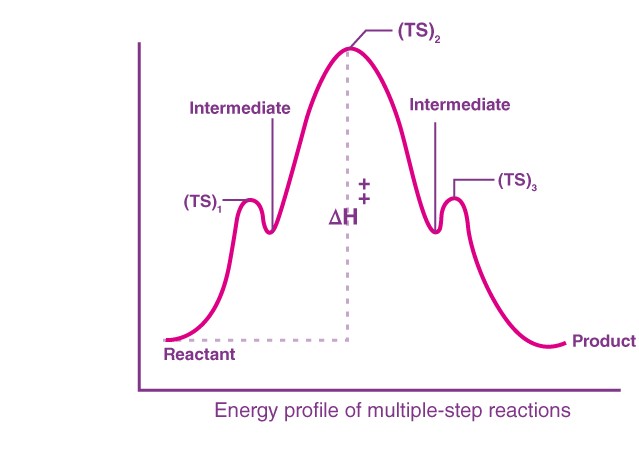
We can see that B and C are the products of a reaction, and hence they are termed intermediates. The highest energy of a particular reaction should be the transition state.
Conclusion:
From the above example, we can show that the intermediates are isolable, that is, they can be isolated. On the other hand, the transition state is not isolable because we assume the reaction to take place via a transition state cannot be isolated.
RELATED POSTS
View all
