
Inductive Effect
It is an electron delocalisation effect via σ bonds that arises due to the difference in electronegativities. For example, in a σ bonded organic compound like C-C-C-Cl, the carbon attached to the chlorine atom can be referred to as the α-carbon, and the one adjacent to that carbon as the ß-carbon and so on.
Now, since chlorine is more electronegative than carbon, it withdraws the electrons that are present via the σ bond toward itself, thereby making Cα fractionally positive. Since it is devoid of electrons, Cα, now being slightly electropositive than Cß, pulls the sigma-bonded electrons of Cα-Cß bond toward itself, and in this process, it makes Cß slightly electropositive.
The electron-withdrawing effect of the chlorine atom is transmitted through the carbon chain via the σ bonds. This transmission of charges decreases rapidly with the number of intervening σ bonds. We can practically ignore this effect beyond Cß.
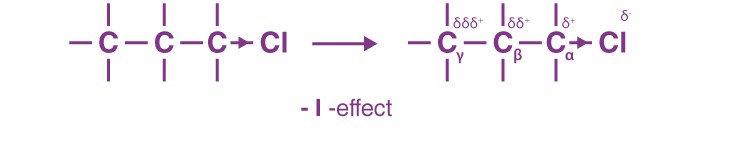
The arrow is pointed more towards the electronegative atom. If a group withdraws the electron from carbon, it makes carbon slightly electropositive. Such groups are called -I groups, and the effect is termed -I effect. For example, -Cl, -Br, -CN and -NO2 are -I groups.
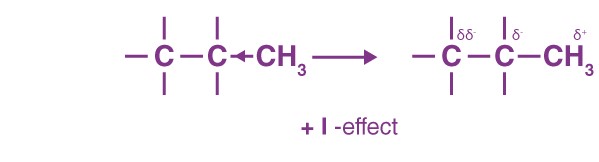
Groups that release electrons towards carbon are termed +I groups, and the effect is termed as +I inductive effect in organic chemistry. For example, alkyl groups like -CH3 are +I groups.
Electromeric Effect
It is the temporary delocalisation of π-electrons in a compound containing multiple covalent bonds. It is important to note that it is only a temporary effect, that is, it occurs only when a reagent is added. The Electromagnetic Effect in organic chemistry can be classified into two types:
- Positive Electromeric Effect
- Negative Electromeric Effect
Positive Electromeric Effect
When the π-electrons are given to the attacking reagent, for example, the reactions alkenes and alkynes mostly occur via +E, this reaction is also called electrophilic addition.
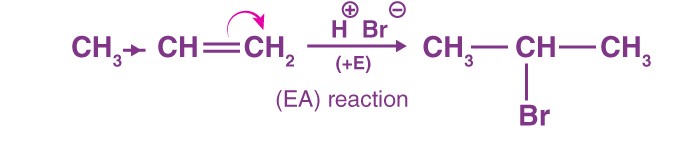
The proton adds at C-1 as the π-electrons were given to the attacking reagent (H+). This results in the formation of a carbocation.
Negative Electromeric Effect
When the π-electrons are shifted to a more electronegative atom (O, N, S) joined via multiple bonds, it is called the negative electromeric effect. For example, the reactions of aldehydes and ketones occur predominantly by the -E effect. It is also called nucleophilic addition.
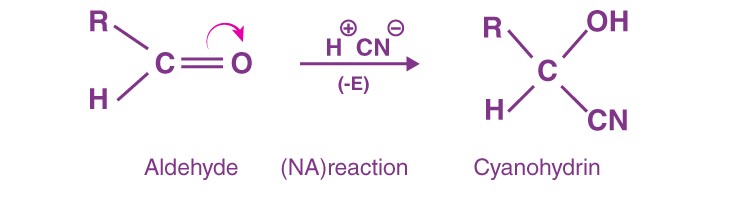
The CN- ion adds to the C atom of the carboxy group opposite to the movement of the π-electron cloud.
RELATED POSTS
View all
