
The stability of carbanions can be explained using the inductive effect.
CH3 — CH2(CH3) — CH(CH3)2 — C(CH3)3 i.e. Methyl Primary (1°) Secondary (2°) Tertiary (3°)
Since alkyl groups are electron-releasing by nature through induction, we can say that the more the number of methyl groups attached to the carbon having a negative charge, the less its stability would be.
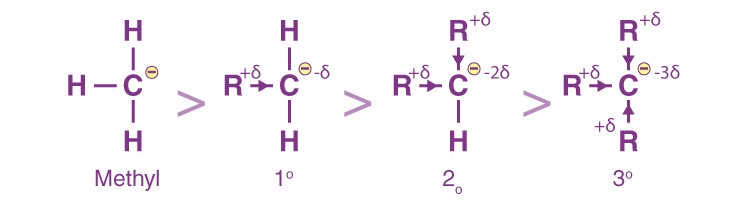
This is because the carbon already has a negative charge, to which the methyl groups push electrons via induction. This results in inter-electronic repulsions and destabilises the species.
Therefore, the stability order for carbanions is as follows:
3°< 2°< 1°< Methyl carbanion
Stability of Free Radicals
The stability of free radicals in organic chemistry follows the same trend as that of carbocations.
CH3–CH2(CH3)–CH(CH3)2–C(CH3)3 i.e. Methyl Primary (1°) — Secondary (2°) — Tertiary(3°)
Therefore, the stability order of free radicals is of order: 3° > 2° > 1° > methyl carbocation.
This can be explained with the help of the Hyperconjugation that we saw, but there would be an overlap between the σ-bond of C-H and the odd electron in the p-orbital of carbon.
RELATED POSTS
View all
