
Carbocations
The stability of carbocations can be explained by the Inductive Effect.
CH3++CH2(CH3) +CH(CH3)2+C(CH3)3, i.e., Methyl Primary (1°) Secondary (2°) Tertiary(3°)
We know that alkyl groups of organic chemistry are +I groups, that is, they release electrons through the sigma bonds.
Since the carbon is deficient in electrons, we can say that as the number of methyl groups increases, the stability of the carbocation increases, as the electropositive carbon is satiated by the electrons given by the methyl groups via the +I effect.
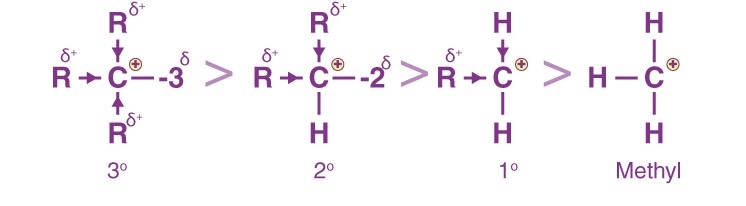
Therefore, the stability order of carbocations in organic chemistry is of the order 3° > 2° > 1° > Methyl carbocation.
Hyperconjugation
When a C-H σ-bond is in conjugation with a carbocation, this effect is observed. A carbocation has a vacant p-orbital. The bonded σ-electron pair of the C-H bond is displaced towards the vacant p-atomic orbital. This increases the electron density in the empty p-AO.
It is, therefore, a resonance effect, where a C-H bond breaks and the σ-electron pair is delocalised to the vacant p-AO of the carbonation. Since the bond between C and H is broken, it is also called ‘no bond resonance’. It is also referred to as the Baker-Nathan effect.
RELATED POSTS
View all
