Osmotic solutions (Tonicity)
May 13, 2024 | by Bloom Code Studio

- Tonicity is the ability of extracellular solutions to induce the movement of water in and out of a cell by the process of osmosis.
- The tonicity of a solution is determined the concentration of solute and solvent molecules in the solution.
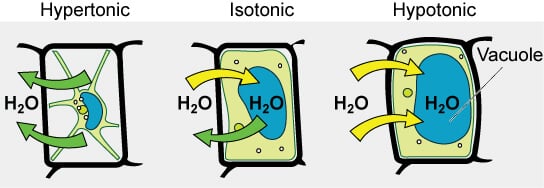
On the basis of the tonicity of solutions, they are termed as hypotonic, hypertonic, or isotonic solutions.
Hypotonic solution
- If an extracellular solution has less concentration of solute than that inside the cell, the solution is termed a hypotonic solution.
- When a cell is placed in a hypotonic solution, the movement of water occurs into the cell resulting in endosmosis.
- The cell is such a condition will swell up and might even burst.
Hypertonic solution
- If an extracellular solution has more concentration of solute than that inside the cell, the solution is termed a hypertonic solution.
- When a cell is placed in a hypertonic solution, the movement of water occurs out of the cell resulting in exosmosis.
- The cell shrinks down, losing the ability to divide, and even function.
Isotonic solution
- When an extracellular solution has the same concentration of solute as that inside the cell, the solution is termed an isotonic solution.
- When a cell is placed in an isotonic solution, no movement of water occurs across the cell membrane.
- In this case, the size of the cell is not influenced as no movement of water takes place.
RELATED POSTS
View all
