Chemistry of Cleansing Agents in Everyday Life
April 28, 2024 | by Bloom Code Studio

What are soap and detergents?
Soaps are sodium or potassium salt of higher carboxylic acid such as stearic acid, palmitic acid and oleic acid, whereas detergents contain a long chain of alkyl groups. Detergents, in comparison to soaps, can also function in hard water.
Saponification: Alkaline hydrolysis of triesters of glycerol to form soap is known as saponification. Soap does not function in hard water since they precipitate in it.
How do soaps work?
Soaps are generally sodium or potassium salts of long-chain fatty acids. Soap molecules have a hydrophobic as well as a hydrophilic part. While the hydrophilic part clings to the water when washing, the hydrophobic end clings to the dirt particles. Thus, when we pour away the water, the dirt particles wash away with the soap molecules.
Types of Soaps
- Toilet Soaps: Potassium soaps are softer than sodium soaps.
- Floating Soaps: They can be prepared by beating soap bubbles.
- Transparent Soaps: They contain soap dissolved in excess of alcohol, and it is evaporated.
- Medicated Soaps: They contain soaps by adding little amounts of Dettol, Savlon, etc.
- Laundry Soaps: They mainly contain sodium rosinate and borax.
Types of Detergents
Anionic Detergent: In this type, anions act as detergents. For example, sodium lauryl sulphate
Cationic Detergent: In this type, cations act as detergents. For example, cetyl trimethyl ammonium bromide.
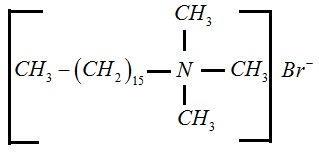
Non-ionic Detergent: They are neutral. The whole molecule acts as a detergent. For example, polyethylene glycol stearate.
Chemistry of Cosmetics in Everyday Life
Cosmetics contain the following categories of chemicals:
- Emulsifiers: They increase the stability of the emulsion. For example, potassium cetyl sulfate.
- Preservatives: They are added to cosmetics to increase their shelf life. For example, benzyl alcohol and salicylic acid.
- Thickeners: They give an appealing consistency. For example, cetyl alcohol and stearic acid.
- Emollients: They soften the skin by preventing water loss. For example, glycerine and zinc oxide.
- Glimmer and Shiners: For example, mica, bismuth oxychloride.
RELATED POSTS
View all
