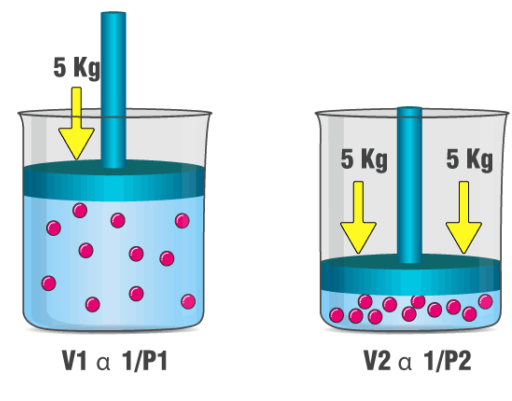
Also known as the names of Boyle–Mariotte law or Mariotte’s law, Boyle’s law states that:
| The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged within a closed system. |